Electricity in general
I quite nearly failed physics when it came to the electricity unit, so this is my second attempt at a very general understanding of it all.
Pretty good visual demonstration: https://www.youtube.com/watch?v=4Q7s7QANevA
These people explain things using a consistent plumbing metaphor: http://science.howstuffworks.com/environmental/energy/question501.htm
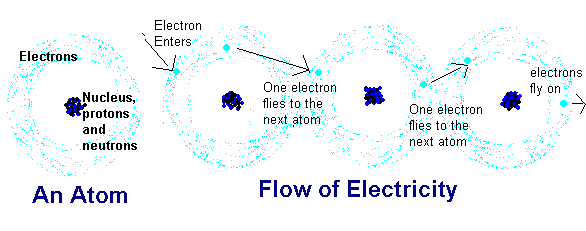
First, you have the atom, with a nuclei of positively charged protons and neutrons, which have no charge. Electrons have a negative charge, and they sort of spin around the nucleus. Atoms with equal numbers of protons and neutrons are overall neutral. If they gain or lose an electron, they gain either a negative or positive charge, respectively, and are considered ions.
Power: The amount of work done over time, the unit is the watt.
Work: The transfer of energy, the unit is the joule.
Energy: The ability to do work.
Joule: The amount of energy necessary to displace a force of one newton by one meter. In terms of electricity, it is the energy transference needed to move an electric charge of one coulomb through one volt, or to produce one watt (=1 joule/second) of power for one second.
Newton: The amount of force needed to give a one kilogram mass an acceleration of one meter per second square. It's about the same weight (downward force) of an apple.
Coulomb: The charge that passes a single point, carried by a current of one ampere, over the course of one second. That's about equal to the cumulative charge of 6.242E18 electrons, a lot of electrons.
Voltage: From the second link, if you think about plumbing pipes as a representation of electrical systems, "voltage is equivalent to the water pressure." This is measured in volts. From below, the units seem to be joules per coulomb.
Current/amperage: "The current is equivalent to the flow rate." This is measured in amperes, or amps for short. If you places two conductors (wires) parallel to each other at a distance of one meter, an amp is the amount of current you would need to run through those wires to "produce a force equal to 2E7 newtons per meter". One amp of current is one coulomb of charge carried past a point per second; the units of current are coulombs per second.
Resistance: "The resistance is like the pipe size." This is measured in ohms. Resistance is voltage over current, which is equivalent to (joules/coulomb)/(coulombs/second). I am not sure what to make of that. One ohm is the resistance needed to produce a current of one amp in a conductor when the voltage across it is one volt.
Pretty good visual demonstration: https://www.youtube.com/watch?v=4Q7s7QANevA
These people explain things using a consistent plumbing metaphor: http://science.howstuffworks.com/environmental/energy/question501.htm
First, you have the atom, with a nuclei of positively charged protons and neutrons, which have no charge. Electrons have a negative charge, and they sort of spin around the nucleus. Atoms with equal numbers of protons and neutrons are overall neutral. If they gain or lose an electron, they gain either a negative or positive charge, respectively, and are considered ions.
Power: The amount of work done over time, the unit is the watt.
Work: The transfer of energy, the unit is the joule.
Energy: The ability to do work.
- Potential Energy: Stored energy that can be converted into kinetic energy, measured in joules. It is determined by an object's relative position and mass. Batteries are used as an example of and a metaphor for potential energy.
- Kinetic Energy: As an object changes position, potential energy is converted into kinetic energy. It is determined by an object's velocity and mass, and also measured in joules.
Joule: The amount of energy necessary to displace a force of one newton by one meter. In terms of electricity, it is the energy transference needed to move an electric charge of one coulomb through one volt, or to produce one watt (=1 joule/second) of power for one second.
Newton: The amount of force needed to give a one kilogram mass an acceleration of one meter per second square. It's about the same weight (downward force) of an apple.
Coulomb: The charge that passes a single point, carried by a current of one ampere, over the course of one second. That's about equal to the cumulative charge of 6.242E18 electrons, a lot of electrons.
Voltage: From the second link, if you think about plumbing pipes as a representation of electrical systems, "voltage is equivalent to the water pressure." This is measured in volts. From below, the units seem to be joules per coulomb.
Current/amperage: "The current is equivalent to the flow rate." This is measured in amperes, or amps for short. If you places two conductors (wires) parallel to each other at a distance of one meter, an amp is the amount of current you would need to run through those wires to "produce a force equal to 2E7 newtons per meter". One amp of current is one coulomb of charge carried past a point per second; the units of current are coulombs per second.
Resistance: "The resistance is like the pipe size." This is measured in ohms. Resistance is voltage over current, which is equivalent to (joules/coulomb)/(coulombs/second). I am not sure what to make of that. One ohm is the resistance needed to produce a current of one amp in a conductor when the voltage across it is one volt.
There is an electric field, and anything on it has electric potential (called a volt (V)). However, one cannot measure the potential of one charge, only the electric potential difference between two charges (this is voltage). This is equivalent to the change in potential energy divided by a positive test charge, which is then equal to the work done divided by the positive test charge. This is not so important, and I do not understand it. Only, the resultant units from the former equivalency are joules per coulomb.
Conveniently, my textbook gives me a voltage example involving batteries. Say you have a 12V battery, meaning that there is a a difference in potential energy of 12 joules per coulomb, and that you have a light hooked up to it. The amount of potential energy turned into kinetic energy (how the light functions) depends on how long you keep the light on. If you leave the light on twice as long, twice as much energy is going to be converted. The total energy transformed (in joules) is equal to the voltage of the system (in volts) multiplied by the amount of charge carried in the circuit (in coulombs).
Conveniently, my textbook gives me a voltage example involving batteries. Say you have a 12V battery, meaning that there is a a difference in potential energy of 12 joules per coulomb, and that you have a light hooked up to it. The amount of potential energy turned into kinetic energy (how the light functions) depends on how long you keep the light on. If you leave the light on twice as long, twice as much energy is going to be converted. The total energy transformed (in joules) is equal to the voltage of the system (in volts) multiplied by the amount of charge carried in the circuit (in coulombs).

All those terms and SI units get jumbled up in my head, but ultimately, the specificity to a better understanding of the greater whole.
Going with the plumbing comparison from above, it's easy to see the relation between current, voltage, and resistance, and how one gets the equations to the left. If you increase the water pressure (voltage), the flow rate past a single point (current) is going to increase. If you increase the diameter of the pipe (resistance), there is going to be more water going past that point (current) as well.
Batteries are, on a basic level, composed of a cathode (the positive side), an anode (the negative side), and electrolytes. Purified water is not conductive, you need minerals/salt for it to carry a charge. Electrons will flow from the anode to the cathode when the circuit is conmplete because the metal that composes the cathode has a lower atomic number than the anode material. This means that there are few electrons in the outer shell of the former, and if given the opportunity, electrons will travel through the electrolyte to get to equilibrium. Batteries attached to solar panels can be recharged by connecting the terminals to a power soure. This gets the electrons to flow back contrary to their normal migration.
links/practical matters
Watt hour usage calculator: https://www.wholesalesolar.com/solar-information/start-here/offgrid-calculator#applianceTable
Watt hours system calculator: https://www.wholesalesolar.com/solar-information/battery-bank-sizing
Watts to amps calculator: http://www.rapidtables.com/calc/electric/Watt_to_Amp_Calculator.htm
Very informative: www.linkedin.com/pulse/amp-hours-vs-kilowatt-oluwaseun-ibosiola
This too: www.batterystuff.com/kb/articles/solar-articles/solar-info.html
I get ~4.2 sun hours a day in the summer, ~2-3 hours in the winter.
To charge two small appliances (estimated to pull 8 watts) for two hours each, and a laptop (estimated to pull 150 watts) for three hours a day, I would need 482 watt hours per day (per charge cycle). This is because (2 appliances * 8 watts * 2 hours) + (1 laptop * 150 watts * 3 hours) = 482 watt hours (watts * hours). The site says, to account for battery inefficiency, assume 626.60 watt hours are necessary, or 0.626kwh.
I'm looking at a battery with a 35 amp hour rating, assuming it's at a standard 20 hour rating, it can provide 1.75 amps per hour.
To find how many watt hours your battery will give, multiply the amp hour rating by the voltage.
Regular car batteries aren't ideal (might not work at all?) for solar energy systems because only they give an initial kick of energy to get things started. If you want to charge a appliances/devices, you'll be looking for a deep-cycle battery (if it's sealed, it needs less maintenance), which steadily releases energy, and can afford to drain more than a regular battery. Unlike a phone, it seems it's a pretty bad idea to use all the charge stored in a car battery.
Watt hours system calculator: https://www.wholesalesolar.com/solar-information/battery-bank-sizing
Watts to amps calculator: http://www.rapidtables.com/calc/electric/Watt_to_Amp_Calculator.htm
Very informative: www.linkedin.com/pulse/amp-hours-vs-kilowatt-oluwaseun-ibosiola
This too: www.batterystuff.com/kb/articles/solar-articles/solar-info.html
I get ~4.2 sun hours a day in the summer, ~2-3 hours in the winter.
To charge two small appliances (estimated to pull 8 watts) for two hours each, and a laptop (estimated to pull 150 watts) for three hours a day, I would need 482 watt hours per day (per charge cycle). This is because (2 appliances * 8 watts * 2 hours) + (1 laptop * 150 watts * 3 hours) = 482 watt hours (watts * hours). The site says, to account for battery inefficiency, assume 626.60 watt hours are necessary, or 0.626kwh.
I'm looking at a battery with a 35 amp hour rating, assuming it's at a standard 20 hour rating, it can provide 1.75 amps per hour.
To find how many watt hours your battery will give, multiply the amp hour rating by the voltage.
Regular car batteries aren't ideal (might not work at all?) for solar energy systems because only they give an initial kick of energy to get things started. If you want to charge a appliances/devices, you'll be looking for a deep-cycle battery (if it's sealed, it needs less maintenance), which steadily releases energy, and can afford to drain more than a regular battery. Unlike a phone, it seems it's a pretty bad idea to use all the charge stored in a car battery.